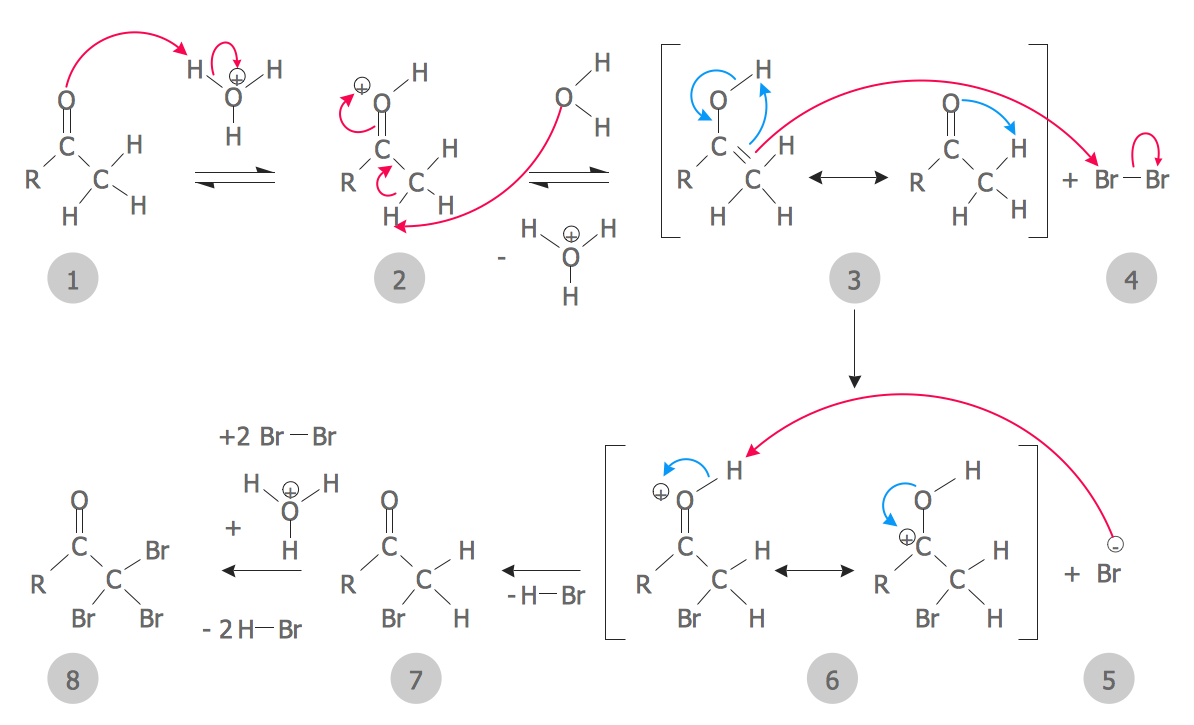
These starting materials, cis-But-2-ene and trans-But-2-ene, which differ only in the configuration of the double bond, lead to stereoisomeric products. What’s even more interesting is that the stereochemistry of the starting alkene is directly related to the stereochemistry of the product.įor instance if we treat cis-2-butene with Br 2, we get a mixture of enantiomers.īut if we treat trans-2-butene, we only get a single product (“meso” 2,3-dibromobutane), which is itself a diastereomer of (S,S)-2,3-dibromobutane and (R,R)-2,3-dibromobutane. Bromination of Alkenes Observation #2: The Reaction Is Stereospecific For instance, in the reaction of cyclohexene with Br 2, the two bromine atoms add to opposite faces of the alkene (“anti” stereochemistry). Possibly the most interesting feature of this reaction is that the products follow a very predictable stereochemical pattern.
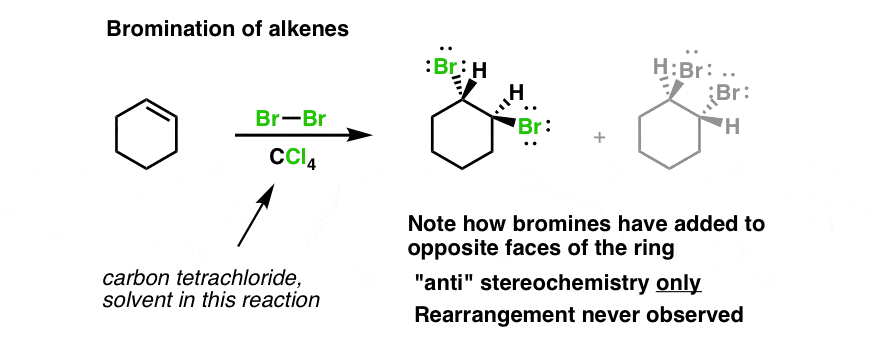
Bromination Of Alkenes Observation #1: Only anti Products Are Observed
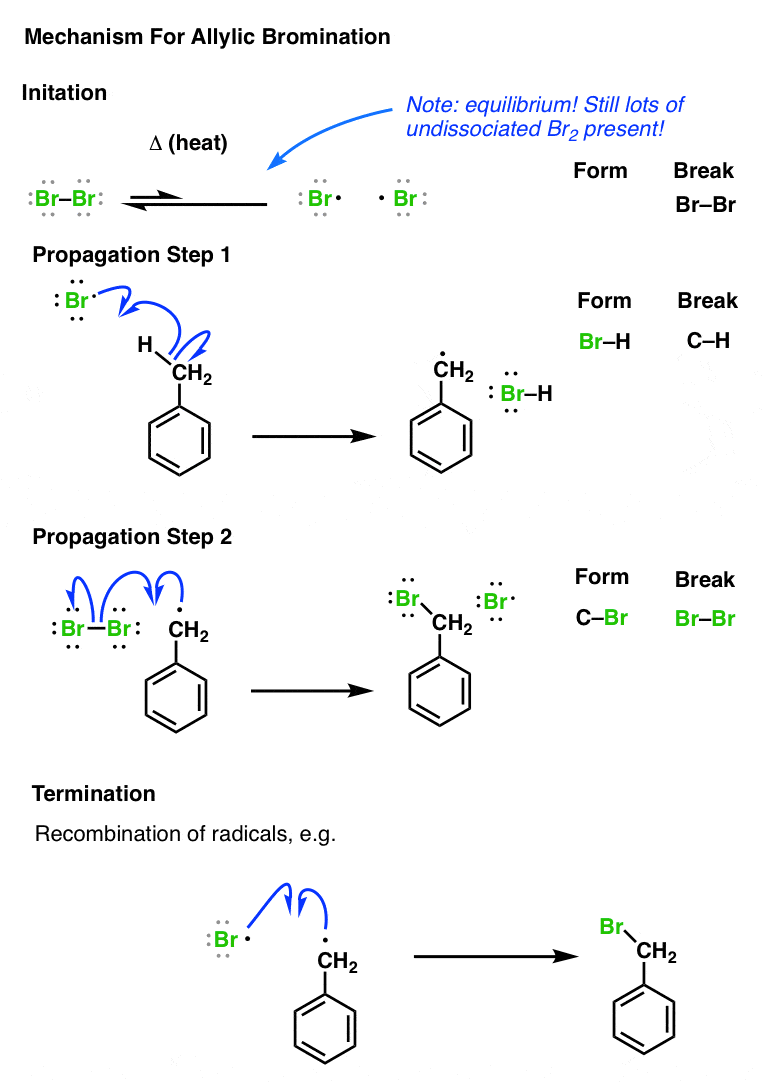
#Bromination mechanism chem draw free
It’s a family of reactions which proceed through 1) attack of an alkene upon an acid, forming a free carbocation, and 2) attack of a nucleophile upon the carbocation.Īlthough we saw that several key reactions of alkenes were consistent with this mechanism, it isn’t the case for all.

In a previous post we went through the key reactions of the carbocation pathway. of #"CH"_3"Br"# forms from the reaction of a methyl radical with a bromine radical.Bromination of Alkenes Gives anti Products of bromine radicals react to reform #"Br"_2#. That is a good situation where they would react with each other, and the termination steps account for that possibility. This amount is small simply because in the actual propagation steps, it is depicted that the methyl radical that forms is supposed to react again with #"Br"_2#, and so if the amount of each reactant is such that a reaction goes partway to completion, you could get a few extra methyl radicals that haven't reacted with anything else yet. of each reactant, there is a chance that two methyl radicals will react in the termination steps, and at that point, you will form ethane. Now, since at some point in the propagation steps, a methyl radical gets formed, if you have enough equiv. of bromine radical in the reaction vessel, which is the bare minimum for the first termination step. of #"Br"_2# reacts with the methyl radical to form one equiv. of a methyl radical and #"HBr"#.Īnother equiv. Then, one of them reacts with methane to heterolytically cleave it and form one equiv. Tracing this mechanism, the initiation step homolytically cleaves the #"Br"-"Br"# bond, giving two bromine radicals. These steps in the mechanism are not about keeping track of how many molecules are unreacted or reacted, but about tracking what kinds of key steps occur that describe the key points of the reaction. Something to keep in mind here is to not get so caught up about the actual equiv. I rectangled where the ethane gets produced.
